 This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. 
Table.A vertical column in the periodic table. We can also find the valency of neon with the help of the periodic So that they do not have any tendency to combine with other elements which Not require to lose or gain electrons to complete its energy shell i.e. Noble gases like neon have two valence electrons so it does Possess both positive and negative valency, and atoms having eight outermostĮlectrons, valency will be zero (i.e. Outermost shells is between one to four, the atom has positive valency and ifĮlectrons are between four to eight, the valency is calculated by subtractingįrom eight and valency will be zero. Lost, gained, or shared with another atom at the time of bond formation.Īn atom is said to be stable when its outermost shells haveĮight electrons (except H & He). The valency of an atom is determined based on the number of electrons Valenceĭescribes how easily an atom or a free radical can combine with other chemical There are many different ways to find out the valency of anĪtom which reflects the ability of an atom to bond with other atoms. Of an atom is called valence electrons, and there is only one electron present The total number of electrons present in the valence shell Of n, and in 2s☢p⁶, the highest value of n is 2 so that the valence shell of Ne The highest number of principle quantum numbers which is expressed in the term Neon electron configuration Ne (10) = 1s 2 2s☢p⁶ (completeĪs we know, the valence shell of an atom can be found from Again another two electrons will go in 2s orbital and the remaining 6 will go to the 2p orbital. the first two electrons will go in the 1s orbital as the S orbital can hold a maximum of 2 electrons only. The neon atom has a total of 10 electrons so, we have to put 10 electrons in orbitals. Less) in any condition for a particular atom and may or may not be equal to itsĮlectron configuration is the arrangement of electrons on The valenceĮlectron for a neutral atom is always definite, it cannot be varied (more or 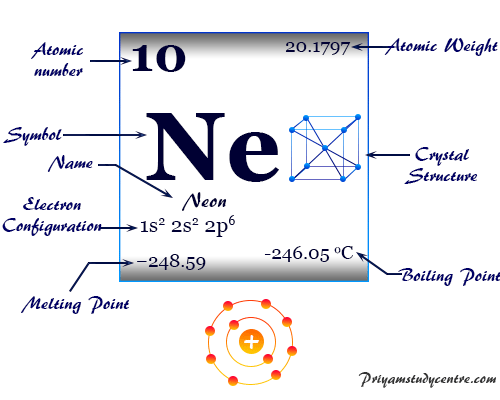
Valence electrons are the total number of electrons present You? But before that let's have some ideas about what these terms are: Difference Between Valence Electrons and Valency 
You are here to know valence electrons and valency of neon, aren’t Indicators, fluorescent lamps, and as a cryogenic in refrigerators. Neon-20, neon-22, and neon-21Īre the most stable isotopes of neon. Asįor now, It has more than 12 isotopes discovered. Till now, not any stable compounds of neon is discovered. It is widely used in fluorescent lamps and electric The second lightest noble gas after hydrogen and glows reddish-orange on the Is colorless, odorless, tasteless, and lighter than air inert gas. Neon, a chemical element with the symbol He and atomic number 10, 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
